
Lead Auditor Training & Certification on MD-QMS Requirements for Regulatory Purposes based on ISO 13485 | TÜV SÜD in India
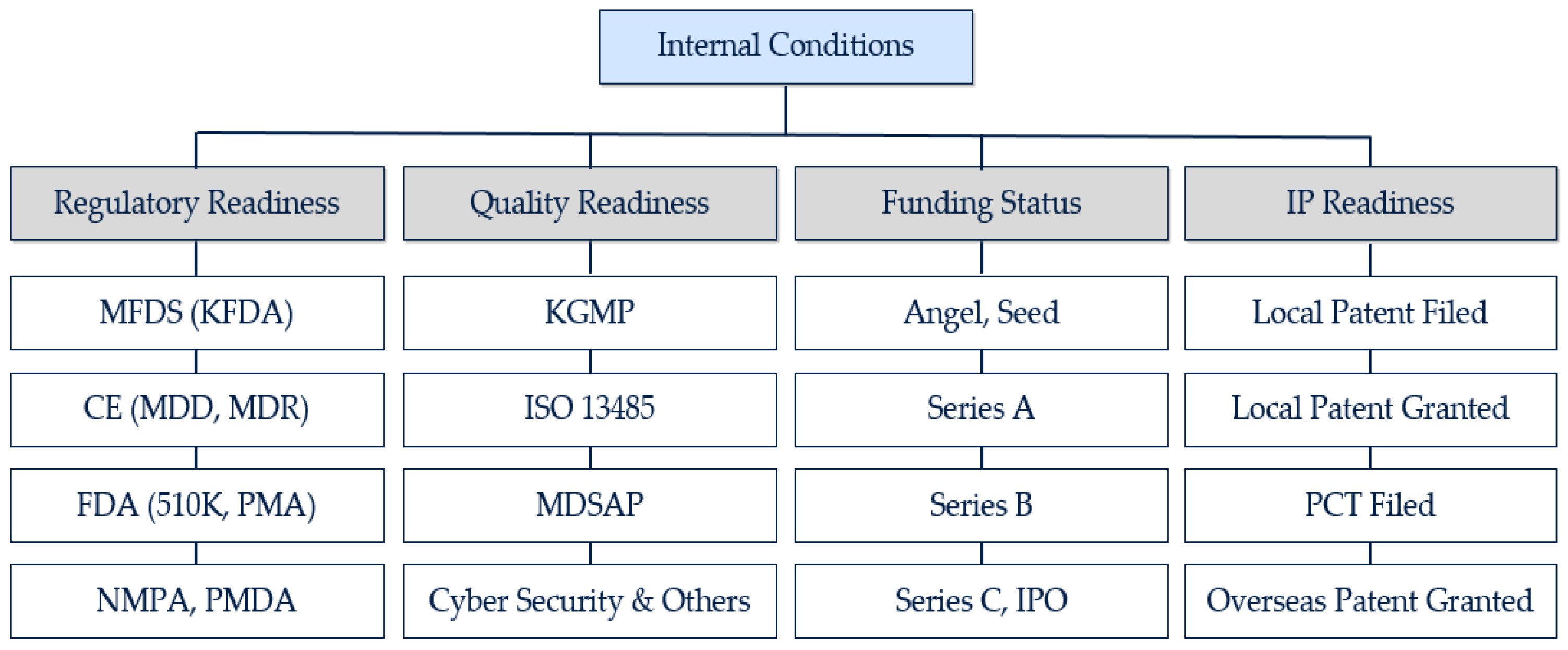
JOItmC | Free Full-Text | What Are the Success Factors for a Partnership with Global Medical Device Companies? Evidence from Korea
IAF Mandatory Document Application of ISO/IEC 17021-1 in the Field of Medical Device Quality Management Systems (ISO 13485) Issu

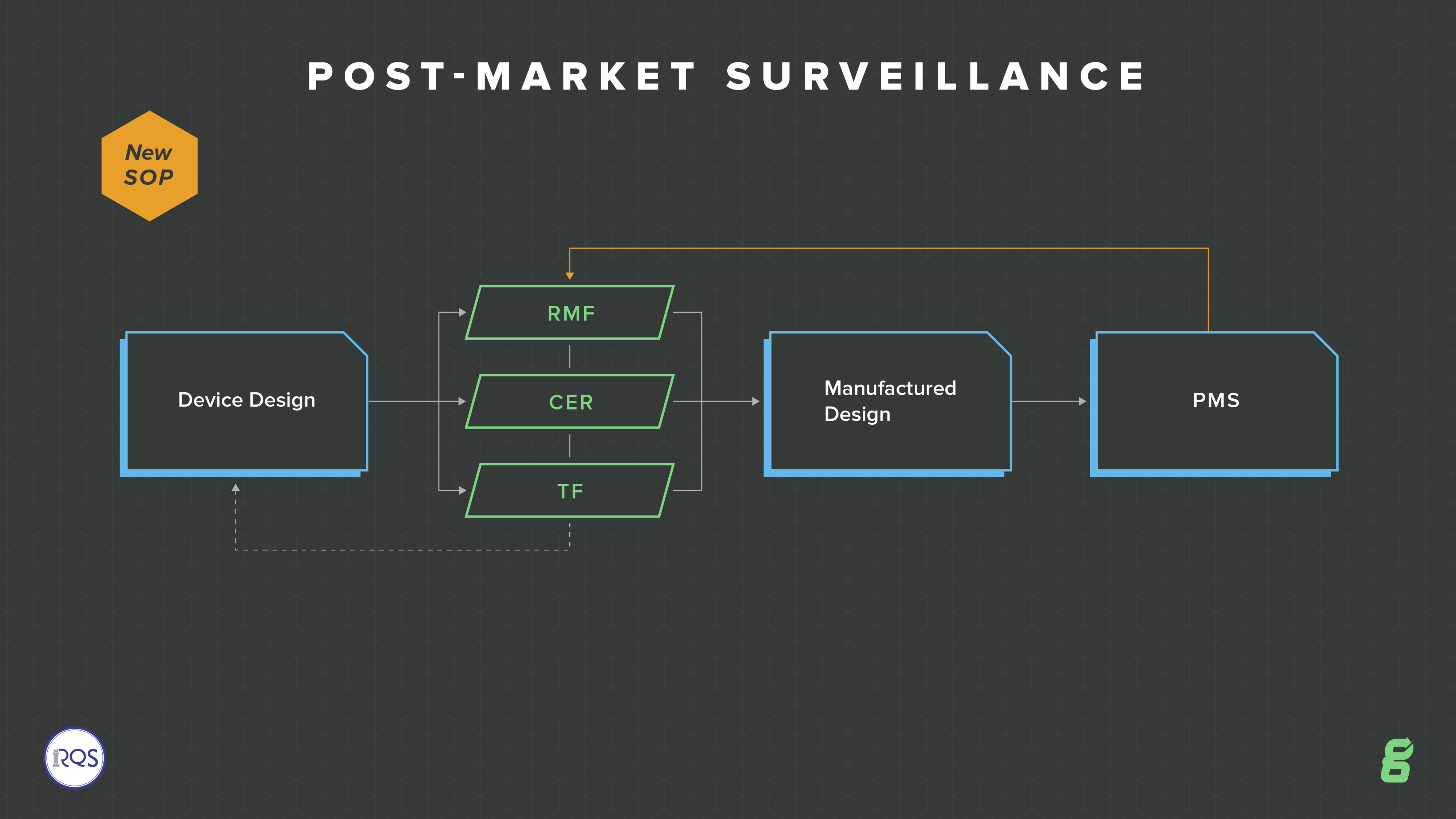
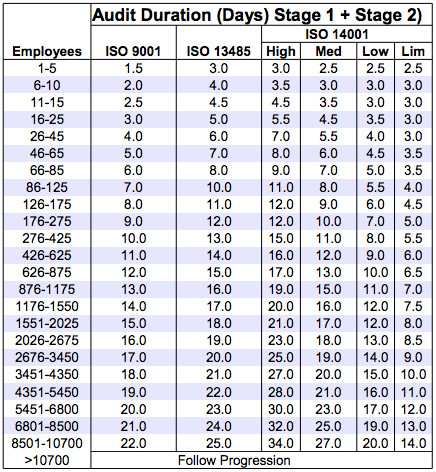

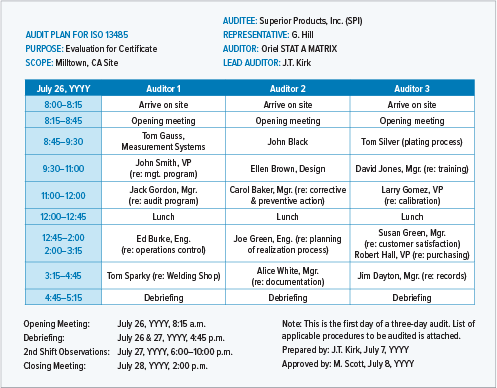

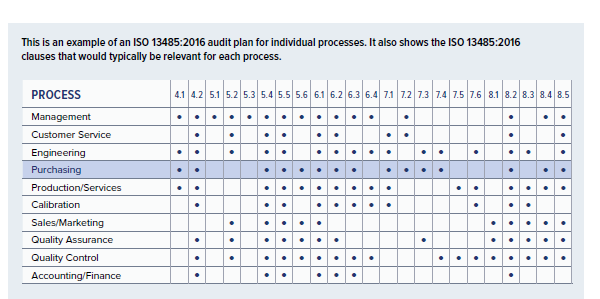


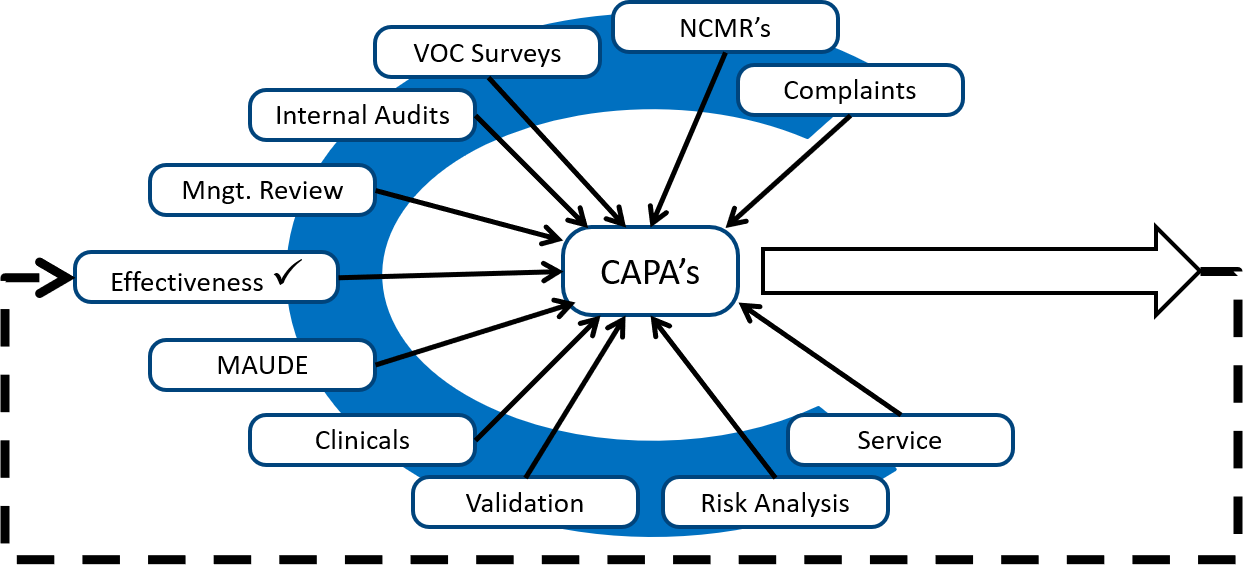






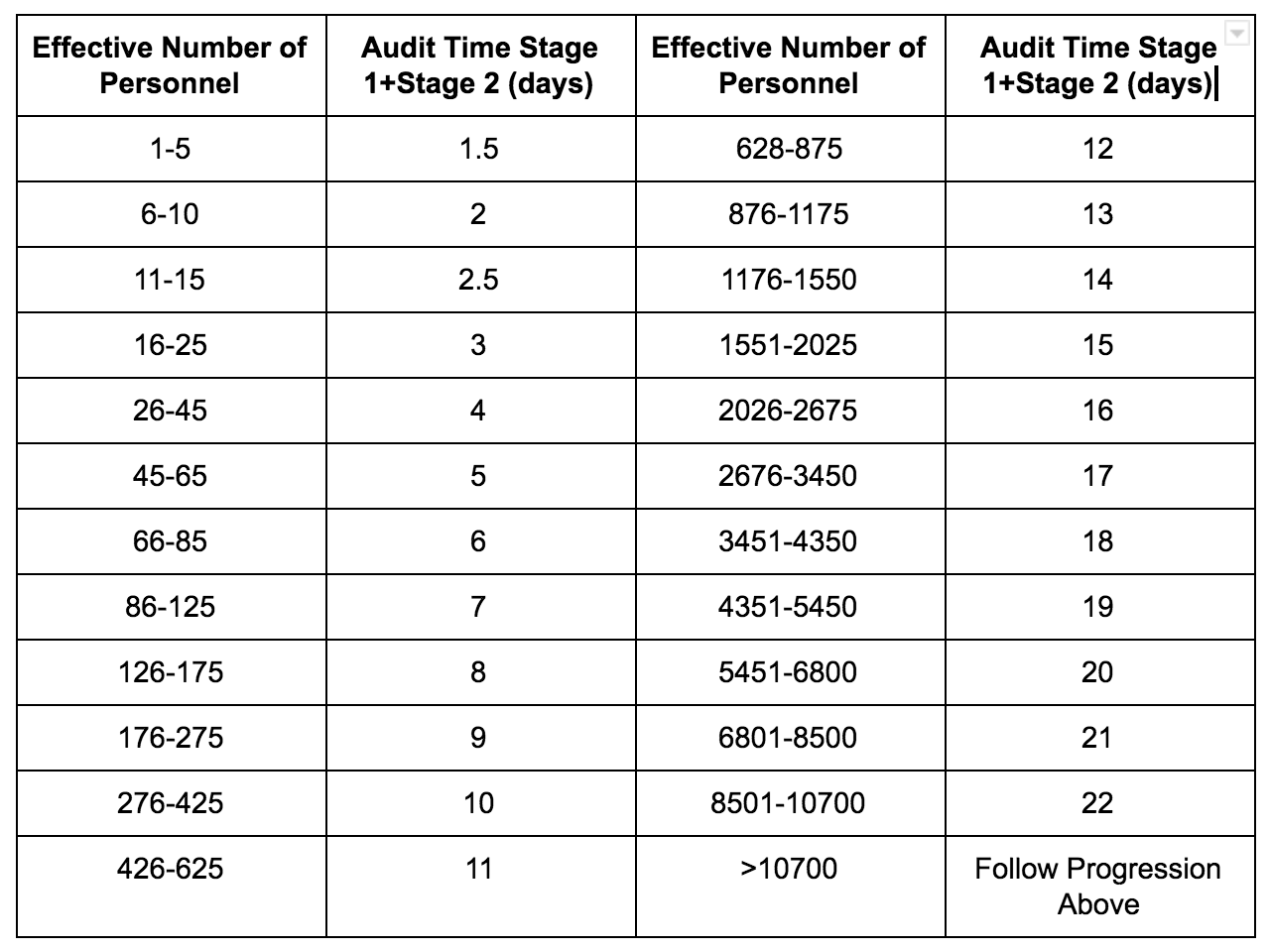
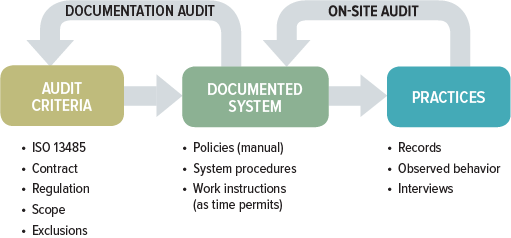


.png?width=250&height=324&name=UG%20ISO%2013485%20(1).png)